BSc 2nd Year Growth and Distribution of Microorganisms Biogeochemical Cycling and Microbial Interactions Notes Study Material
BSc 2nd Year Growth and Distribution of Microorganisms Biogeochemical Cycling and Microbial Interactions Notes Study Material: BSc is a three-year program in most universities. Some of the universities also offer BSc Honours. For BSc, there are certain things that you require the most to get better marks in BSc. Out of those, there are BSc Study Material, BSc Sample Model Practice Question Answers along with BSc Previous Year Papers.
At gurujistudy.com you can easily get all these study materials and notes for free. Here in this post, we are happy to provide you with BSc Growth and Distribution of Microorganisms, Biogeochemical Cycling, and Microbial Interactions Notes Study Material.

BSc 2nd Year Growth and Distribution of Microorganisms Biogeochemical Cycling and Microbial Interactions Notes Study Material
Environmental microbiology quite recently emerged as a new field of study, keeping in view the growing importance of the environmental use of microorganisms. Microorganisms play a key role in environmental processes. Some of their roles are very well-known and worked out. For instance, the role of microbes in the biogeochemical cycling of elements and matter, soil fertility including the decomposition of organic matter, and nitrogen fixation.
Similarly, industrial applications of microbes in medicine, food, acids, vitamins, alcohol production, brewing, and baking are also well known. In these areas, new technologies are being developed and new strains of concerned microbes are being developed by microbial biotechnologists to increase the industrial production of chemicals.
Besides the abovesaid roles of microbes, it has only been recently realized that microbes have the potential of solving serious environmental problems. This potential of microbes can be exploited in suitable ways, and environmental use of microbes should help sustainable development in different sectors.
Microbes have been found to play vital roles in environmental processes, such as degradation and disposal of wastes and pollutants (solid waste; liquid waste such as sewage; pesticides; laundry detergents, oil spills, etc.) and deterioration of materials including items of cultural property and heritage, such as monuments, paintings, etc., and agricultural produce in storage and transit.
Microbes can also be used as biocides in the management of diseases and pests of agriculture, forestry, and public health, a suitable alternative to harmful chemicals. Very recently microbes have also been found to play role in the enhanced recovery of metals, including uranium through bioleaching, from low-grade ores, and in the recovery of petroleum. These recently realized environmental implications and the use of microorganisms developed into a new field of study, environmental microbiology.
This field deals with the possible use of microorganisms in solving environmental problems and helping in environmental management to improve the quality of the environment and life. This should help sustainable development – judicious use of natural resources, making them available to generations to come.
Environmental microbiology includes different fields. These are as follows:
(1) Distribution of microbes in their natural habitats (atmosphere, hydrosphere, lithosphere) including extreme environments, and study of the factors that affect their growth.
(2) Biogeochemical cycling and interactions among microbial populations (various mineral and gaseous cycles of nature).
(3) Agricultural microbiology (soil fertility; diseases and pests; biocontrol of pests: pesticides microbiology i.e. biomagnification and biodegradation).
(4) Environmental quality (biodegradation of wastes and pollutants).
Growth of Microorganisms
Microorganisms represent a significant percentage of the total biomass on the earth. As a result of high rates of their growth and metabolic activity and their adaptability, they are found almost everywhere and occupy an important trophic position in the proper functioning of ecosystems. They are ubiquitous in distribution. Any part of the biosphere supporting the complex, multicellular higher organisms shall contain microorganisms, whereas the converse is not true.
Growth may be defined as the orderly increase in all cellular constituents and results from the biosynthetic and energy-generating processes. The growth may refer to the increase in the mass of a single cell or to the increase in the size of a population of cells. We shall briefly consider both levels of growth of unicellular microbes as well as of multicellular mycelial growth forms such as fungi, and actinomycetes.
Cell Cycle
Unicellular bacteria divide typically by binary fission, in which a cell grows to twice its size and then divides into two identical daughter cells. Each of these cells grows in the manner of the parent cell until it too divides. The sums of all the processes occurring between cell divisions make up the cell cycle. To function efficiently, the cell cycle must involve the controlled regulation and timing of biosynthesis, DNA replication, equal division of nuclear matter, septation, and separation of daughter cells.
In Escherichia coli growing at 37° C in a rich nutrient medium replication of DNA length, almost 1000x the cell length takes only 40 minutes. This part of the cell is called the C-period. Under these conditions, cells will divide, on average, every 20 minutes. Following each C-period, there is a gap of 20 minutes (called the D-period) between the termination of replication and subsequent separation. Consider now a cell growing in a relatively poor medium such that the time between divisions, the doubling time, is 60 minutes.
A newborn cell will contain a complete chromosome that immediately begins replication. After 40 minutes (C-period) the cell will contain two complete chromosomes and after a further 20 minutes (D. period) the cell will divide, segregating the two chromosomes.
The combined length of the C- and D- periods equals 60 minutes, there are no gaps and the cell cycle is said to operate continuously. If the cell, however, is growing in a richer medium which allows a doubling time of 40 minutes, then the C- and D- periods exceed the doubling time and cell cycles must overlap i.e. the D-period of one cycle will overlap with the first 20 minutes of the C-period of the next cycle. Consequently, a newborn cell must contain a chromosome that is already half-replicated.
After 20 minutes it! will contain two chromosomes each of which will begin a new round of replication. When cell division occurs 20 minutes later, each daughter cell will again contain, one hall replicated chromosome. A newborn cell in medium giving a doubling time of 30 minutes should accordingly contain a chromosome that is three-quarters replicated. The cycles must now overlap by 30 minutes.
The eukaryotic cell cycle is usually divided into a division phase (M) and a synthesis phase (S), separated by two ‘gap’ phases (G1 and G2) when DNA is not synthesized. Chromosomal replication proceeds bidirectionally from several, rather than a single site, and cytoskeletal structures are involved in the separation of daughter chromosomes.
Population Growth
We can consider the growth of a population of unicells as an increase in total cell mass-biomass resulting from growth, or as an increase in total cell number resulting from both growth and division. This can be seen in the growth and multiplication of a single cell, and the same results occur in a synchronously dividing population of cells.
However, we normally deal with a large number of cells, as a bacterial colony growing on an agar plate. With such large populations, the chances are that the percentage of cells (not all cells in any population) dividing in any one small period of time will be the same as that in any other period, and if we plot biomass or cell number against time, we will get the same curve.
In other words, the cells in the culture are dividing asynchronously (some cells dividing roughly at the same time period, others at different timings). Consequently, normal growth and multiplication may be taken as synonymous. We can measure any of these, whichever is convenient.
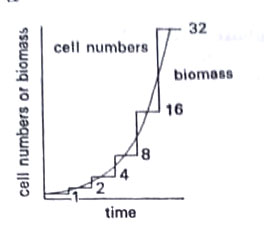
For the study of multiplication, one must measure the actual number of organisms. The cell number is counted under the microscope. Staining methods have been developed to locate live cells. We may obtain a total count of live and dead cells as well as the viable count. A variety of methods have been developed to measure growth as distinct from multiplication. These include the determination of the dry weight, the amount of a typical cell component, etc.
Batch Culture
We will consider what happens when growth is measured after inoculation of a small number of suitable microorganisms into a sterile medium contained in a vessel of a definite volume. A typical growth curve, where the following six main phases may be recognized:
1. Lag phase. This period, in which no growth or multiplication occurs, represents a time when the inoculum cells are adapting themselves to active growth in the new environment. This phase is long if the inoculum cells are old, damaged, or grown previously in a different medium.
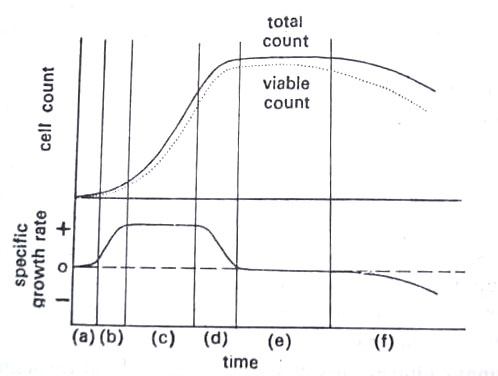
2. Acceleration phase. Normally cells come out of the lag phase asynchronously, giving rise to an acceleration phase where the population growth rate gradually increases as the proportion of cells growing increases. In a biomass growth curve, the lag phase will be shorter, because cells must grow and double in size before they divide and multiply.
3. Exponential phase. Here the cells are growing at a constant rate, that is the generation time (the time between successive divisions) is constant. This is the only phase when the population is growing at a constant rate and it represents the maximum rate and minimum generation time for the prevailing conditions. We have, therefore, autocatalytic growth, with the rate of increase in biomass (x) and the cell number (n) proportional to the existing biomass or cell number. This can be shown by the differential equations given below:
Rate of change of biomass
dx/dt = µx
Rate of change of cell number
dn/dt = µn
where t represents time and µ is the specific growth rate. These equations may be integrated into the following, more practical expressions.
x=xo e µt
n =no e µt
or log x = log xo + µt
log n = log no + µt
where xo and no are values for x and n at the beginning of the exponential phase.
Thus, both biomass and cell number increase exponentially. The consequence is that increase in cell number becomes explosive. Thus in a bacterium with a volume of 1 µm3 growing with a generation time of 20 minutes, after 200 minutes the cell number will increase by a factor of 1024. This means as long as exponential growth continues, there will be a further thousandfold increase every 200 minutes. However, this cannot go indefinitely.
4. Deceleration phase. After 50 hours of incubation, the volume of cells would be greater than that of the whole earth, as a result of exponential growth. This cannot occur in practice and growth must be stopped. The culture, therefore, enters the deceleration phase during which the specific growth rate declines. This decline is generally due to the accumulation of toxic metabolic products or changing environmental conditions, such as reduced pH, etc. In yeasts, growth stops as the ethanol level reaches 10%.
This wine cannot be prepared with an alcoholic content much above 14%. During the deceleration phase, cells often begin to produce secondary metabolites many of which are of industrial value, such as antibiotics.
5. Stationary phase. This phase is brought about through lack of food, or more accurately through exhaustion of one particular nutrient or when an accumulation of toxic products completely inhibits the growth. In either case, the biomass and cell number remain constant.
The cells remain in a state of suspended animation. The biomass and cell numbers produced during this phase depend largely on the initial substrate concentration and on the organism’s, growth yield. This is the mass of cells produced per unit of substrate converted and is often related to the efficiency of growth on a particular substrate. It is therefore of importance in industrial processes where the objective is to produce biomass as cheaply as possible. (Growth & Distribution of Microorganisms Notes Study Material)
6. Death phase. In this phase, the cells begin to die and as a result, there is an increasing divergence between the total and viable counts. However, when death occurs, autolysis takes place i.e. cells begin to digest themselves. Such autolysis has interesting results.
If a laboratory culture is allowed to enter into the death phase, so that a major proportion of cells lyse, those remaining may be able to grow on the products of this lysis. In other words, cannibalism occurs. This short growth phase is followed by a further death phase and the process is repeated until no cells remain. In this way, a culture may remain viable for a very long time, although it should be noted that we are eventually selecting those organisms in a culture that lyse readily or are the best cannibals.
In evolution, microbes have had to compromise between the requirement for maximal growth rates and an ability to adapt rapidly to the environment with a concomitant maximal ability to survive after the onset of the stationary phase. An excellent method of prolonging the stationary phase is to sporulate and produce a cell specialized for the minimal rate of destruction in an environment inimical to cell growth.
Continuous Culture
We have considered so far the batch culture of microbes, where growth occurs in a vessel of finite dimensions and eventually ceases. Is it not possible to devise an apparatus in which the cells are kept growing indefinitely by the continuous addition of fresh nutrient medium and the continuous removal of cells and their products? The work on these lines could be initiated only in the 1940s and much of the development was the result of work on more efficient ways of producing cells for biological warfare (or as we are usually told, for defense against biological warfare).
The method of growth was called continuous culture and the apparatus most commonly used was the chemostat, Fresh sterile medium is pumped into the growth vessel at a steady rate while a constant-level device removes cells and their products at an equal rate. A stirrer homogenizes the fresh medium with the culture and normally also acts as an aeration device. The apparatus may be elaborated further by devices for sampling, pH control, oxygen control, temperature control, antifoam addition, etc.
If we inoculate a chemostat and supply fresh medium at a constant rate, the rate of change in biomass concentration will depend on the increase resulting from growth and the decrease due to the removal of cells in the effluent:
dr / dt = µx – Dx
growth removal
The symbol D represents the dilution rate which is equal to the rate of flow of fresh medium divided by the volume of medium in the culture vessel. If D is less than the maximum specific growth rate of the organism, a steady state is eventually established, in which the actual specific growth rate equals the rate of removal, i.e. µ = D.
This occurs because the specific growth rate becomes limited by the concentration of a single substrate and falls below its maximum value. The equation for the relationship between substrate concentration (s) and specific growth rate (µ) is analogous Michaelis-Menten equation for enzyme kinetics. It has the form:
µ = µms, where µm is the maximum specific growth rate achieved
Ks + s
when the substrate is in excess, and equivalent to the specific growth rate during exponential growth in batch culture. This equation is known as the Monad equation after the French microbiologist who first proposed it. The term Ks is the saturation constant for growth and is the substrate concentration at which µ is 1/2 µm. The smaller the Ks value the greater the organism’s growth rate at low substrate concentration. Bacteria generally have very low saturation constants, reflecting their ability to scavenge the low levels of nutrients often present in nature; this is one factor that determines competitive ability.
The relationship between substrate concentration and specific growth rate as described by the Monad equation can be expressed in the form of a curve in the figure. However, in practice, the curve obtained in such a figure will be slightly different. This is because the cells besides requiring energy for growth, must also maintain their structural integrity and carry out processes not directly associated with growth.
The energy required for such processes is called maintenance energy and if we include it in the relationship the line in any figure designed will not pass through the origin, as even a non-growing cell will require nutrients to supply energy for maintenance.
Chemostats, therefore, operate at sub-maximal specific growth rates where growth is substrate limited. In the steady state, biomass concentration, substrate concentration, and everything else in the vessel remain constant, and in theory, can do so indefinitely.
Continuous culture has many advantages over batch culture, particularly in the research laboratory where reproducibility in the environment is more important than simplicity in technique and also in an industry where large amounts of cells or products are required at economic costs. Some of the advantages are as follows:
(1) By altering the composition of the medium the substrate which is limiting growth may be changed and the specific roles of different nutrients can be studied.
(2) Environmental conditions and specific growth rates may be kept constant over extended periods and not just for the length of the exponential phase.
(3) Growth at submaximal rates may be studied and the growth rate may be changed to a new constant value merely by altering the setting on the medium pump.
(4) Effects of growth rate can be separated from those of environmental factors. In a chemostat, for example, the temperature can be increased or decreased but the actual specific growth rate will be constant as long as D is kept constant. Conversely, we can change D and µ while keeping the temperature constant.
Due to these and other advantages, the chemostat has become a powerful tool in the study of microbial biochemistry, physiology, and genetics and also in our understanding of the ecology of microorganisms and their interactions. However, a chemostat, a relatively sophisticated piece of instrument is to be used properly, as contamination and/or evolutionary change over long periods may be a problem.
Mycelial Growth
The filamentous growth results when cells do not separate after division. This growth form is most highly developed in fungi and filamentous actinomycetes. Here, an extension of individual filaments, or hyphae occurs through the addition of material to the hyphal tips. In most fungi material is synthesized in a length of hypha called the peripheral growth zone and is transported to the tip in membrane-bound vesicles.
Hyphae of Neurospora crassa extend up to 38 um/minute, and have a peripheral growth zone length of 4 mm. Approximately 38000 vesicles must fuse with the tip every minute to maintain this extension rate. Materials synthesized behind the peripheral growth zone will never catch up with the tip. The materials are thus to be utilized elsewhere, as in the initiation of a branch.
The repetition of this process eventually leads to the development of a complex branching mycelium. Since cells do not separate in filamentous organisms, the cell cycle can not be defined. Instead, there is a duplication cycle, in which the apical cell doubles its length, nuclear division takes place, septation separates the daughter nuclei and a branch is formed.
In a sense, branch production is equivalent to cell division and mycelial organisms grow exponentially. In liquid culture, filamentous forms give a batch growth curve similar to that and also can be grown successfully in continuous culture.
The filamentous forms are adapted to the colonization of solid substrates and surfaces. Unicells typically form small, self-limiting colonies on agar plates while a single fungal colony may cover the whole plate. This is because filamentous organisms can regulate the distribution of biomass between tip extension and branch formation. (Growth & Distribution of Microorganisms Notes Study Material)
On nutrient-poor media, branch production is expressed so that material may be directed towards hyphal tips to maintain rates of extension into regions of fresh nutrients. On nutrient-rich media, branch production is increased to enable more extensive colonization behind the growing margin. Unicells cannot regulate biomass production in this way and colonies are formed by cells pushing each other aside and piling on top of one other.
Synchronous Growth
A growing microbial culture contains cells dividing asynchronously and the properties of the population are the average properties of the individual cells. While studying cell cycle events we want to measure changes in the biochemical features of individual cells and we, therefore, need to amplify the physiological events by producing a synchronously dividing culture in which all the cells divide at roughly the same time. There are two methods available for producing synchronous cultures.
1. Induction methods. They rely on synchronizing an exponential phase culture by appropriate and usually sudden changes in the environment, such as alteration in temperature, the concentration of nutrients, or illumination photoautotrophs.
2. Selection methods. The cells are physically separated from an exponential-phase culture at a particular point in the growth cycle. The methods include (i) centrifugation on a density gradient (ii) filtration of cells through a cellulose nitrate filter and inverting it and passing medium through the filter from above. When cells divide they fall off the filter giving a continuous supply of newly born cells, (iii) the filtration of cells of certain sizes or the selective adsorption of cells on surfaces.
Fed-batch Culture
Although batch and continuous culture are used most frequently in the laboratory and are most useful for experimental work, industrial processes often use a hybrid of the two techniques called fed-batch culture. This involves ordinary batch growth until the substrate is fully utilized, followed by regular additions of the fresh substrate which is then rapidly metabolized.
The total culture volume may be allowed to increase or portions may be removed as the fresh substrate is added. This type of culture was developed empirically but microbiologists are now initiating to understand the physiology of growth under such conditions and the reasons for its efficiency in terms of product formation, yield, and greater control. This may lead to further advances in industrial microbiology.