Microbiology Immune Disorders Notes Study Material
Microbiology Immune Disorders Notes Study Material: BSc is a three-year program in most universities. Some of the universities also offer BSc Honours. After getting enrolled for BSc, there are certain things you require the most to get better grades/marks in BSc. Out of those, there are BSc 2nd Year Study Material, BSc Sample Model Practice Mock Question Answer Papers along with BSc Previous Year Papers. At gurujistudy.com you can easily get all these study materials and notes for free. Here in this post, we are happy to provide you with BSc 2nd Year Microbiology Immune Disorders Notes Study Material.
Microbiology Immune Disorders Notes Study Material
Investigations were carried out on some disorders during the Golden Age of Microbiology. Such disorders, though not so harmful as microbial diseases but were a source of much discomfort and inconvenience. Such diseases were hay fever and local reactions produced by pollen grains on the skin and other membranes. Now we know that hay fever is regarded as a disorder of the immune system and many of us suffer from allergic reactions to foods, cosmetics, leather, metals, and whatnot. Some people can not keep pets due to severe sensitivities.
The common denominator among these problems is a state of increased reactivity known as hypersensitivity. First reported in the early 1900s by several investigators, hypersensitivity stems from the activity of the immune system and involves both humoral and cellular aspects of immunity. This is currently a major area of intense research in immunology. We shall focus in this post on hypersensitivity. The others, including in the broad category of immune disorders, viz. autoimmune diseases, will also be considered here along with the basic concepts of transplantation research and tumor immunology. (Microbiology Immune Disorders Notes Study Material)
Hypersensitivity
Hypersensitivity is a multistep phenomenon that generally requires (i) exposure to an antigen, (ii) a dormant stage during which an individual becomes sensitized, and (iii) a reaction to subsequent exposure to the antigen. The process may involve humoral antibodies, sensitized T-lymphocytes, or sometimes both. A response by antibodies to the subsequent dose of antigens often occurs early, say within minutes. This type is known as immediate hypersensitivity. However, a response by T-lymphocytes usually develops over two or three days and is thus called delayed hypersensitivity. (Microbiology Immune Disorders Notes Study Material)
Gell and Coombs’s classification of hypersensitivity
In the early 1970s, P.G.H. Gell and R.A. Coombs proposed another method of classification of hypersensitivity. Their classification is as follows:
- Immediate hypersensitivity. It includes three types:
(a) Type I (anaphylactic hypersensitivity). It involves lgE, mast cells, basophils, and mediators that induce muscle contraction.
(b). Type II (cytotoxic hypersensitivity). It involves IgG, IgM, complement, and the destruction of host cells.
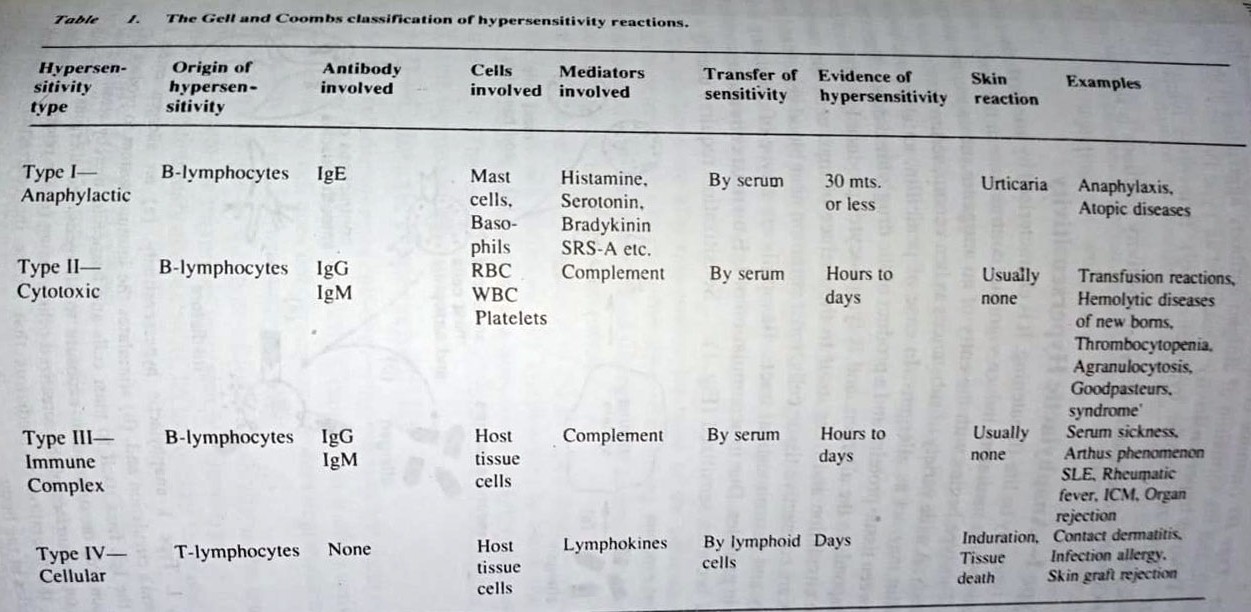
(c) Type III (immune complex hypersensitivity). It involves IgG, IgM, complement, and the formation of antigen-antibody aggregates in the tissues.
- Delayed hypersensitivity. Described as Type IV.
(d) Type IV (cellular hypersensitivity). It involves lymphokines and T-lymphocytes.
Type 1-Anaphylactic Hypersensitivity
This type may be life-threatening. It is characterized by anaphylaxis, a series of events in which mediators induce vigorous contractions in the body’s smooth muscles. This type begins with the entry of an antigenic substance, the allergen, into the body. A wide variety of substances as bee venom, serum proteins, or drugs such as penicillin may act as allergens.
In the case of penicillin, an allergenic complex forms between tissue proteins and a product of the drug’s breakdown. The immune system responds to the allergen, and B-lymphocytes produce IgE. This antibody enters the circulation and fixes itself to the surface of mast cells and basophils. Mast cells are connective tissue cells, most common near blood vessels and in the respiratory and gastrointestinal tracts. Basophils are a type of circulating leukocyte, rich in granules. (Microbiology Immune Disorders Notes Study Material)
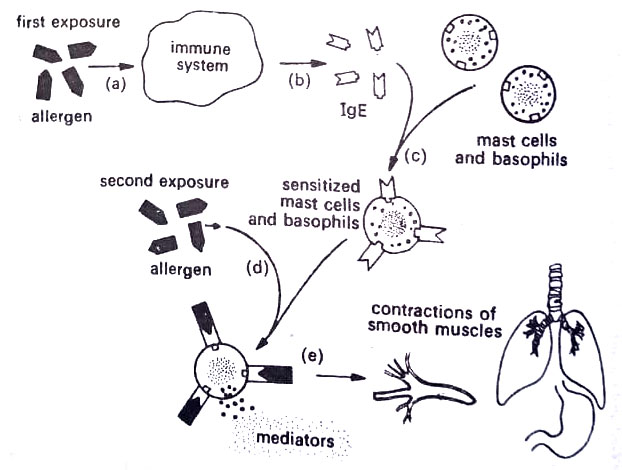
Due to the accumulation of IgE on mast cells and basophils, the individual becomes sensitized. Sensitization requires usually a minimum of one week. Symptoms of anaphylaxis occur rapidly on reexposure to the allergen. Allergen molecules unite with IgE on surfaces of sensitized cells, that are now activated. Mediators such as histamine, serotonin, and Uradykinin are involved.
The next step is degranulation of the sensitized cell, thereby releasing the mediators. Due to this effect, the skin becomes swollen about the eyes, wrists, and ankles, a condition called edema. A person sensitized to an allergen may undergo desensitization, by injecting the antigens to relieve sensitivity to that antigen (allergen),
Atopic diseases
The abovementioned Type I hypersensitivity need not result in whole body involvement that accompanies anaphylaxis. Indeed the vast majority of hypersensitivity reactions are accompanied by limited production of IgE and the sensitization of mast cells in localized areas of the body. The result is an atopic disease or common allergy.
An example of an atopic disease is hay fever. This condition results from inhalations of tree and grass and weed pollens in particular seasons of the year. Immune stimulation by pollen antigens leads to IgE production, and sensitization of mast cells follows in the eyes, nose, and upper respiratory tract. Subsequent exposures bring sneezing, tearing, swollen mucous membranes, and other symptoms.
Allergic reactions are also responsible for most asthmatic attacks. Asthma is characterized by wheezing and stressed breathing and is caused by the same allergens associated with hay fever. Food allergies are accompanied by swollen lips, abdominal cramps, nausea, and diarrhea.
There are two theories to explain how allergies develop. According to one, atopic persons lack sufficient IgA lymphocytes to block the antigens (allergens). In normal individuals, IgA lymphocytes shield IgE lymphocytes from antigenic stimulation. The second theory explains atopic diseases on the basis of a defect in the suppressor T-lymphocytes.
Type II—Cytotoxic Hypersensitivity
This type is a cell-damaging or cell-destroying reaction that occurs when IgG reacts with antigens on the surfaces of cells. Complement is often activated and IgM may be involved but IgE does not participate, nor does degranulation of mast cells. The cells affected are known as target cells.
A well-known example of this type of hypersensitivity is the transfusion reaction that arises from the mixing of incompatible blood types.
Before dealing with transfusion reactions, let us consider the blood groups in humans.
Blood groups -The ABO system
Harvey discovered that repeated attempts to transfuse blood from one individual to another led to disastrous reactions. The reason for this became known only when Landsteiner discovered the alloantigens (alloAgs) of human RBCs in 1900.
AlloAgs are cell surface Ags, i.e. present on surfaces of RBCs. These are Ags encoded by polymorphic genes so that various alleles are present in different individuals from the same species. In the 1890s, there were observations showing that animal species could be distinguished by the reactions of their RBCs and serum proteins with specific antisera.
Landsteiner sought to determine whether individuals of the same species could be distinguished in the same way. When individual samples of serum and RBCs from 22 human species were mixed in all possible combinations the red cells of some persons were found to be clumped by the serum of certain other individuals which led to a classification of the subjects into four groups.
From the results, Landsteiner concluded that
(1) The RBCs of any human carry one, both, or neither of two different Ags (or blood group substances), A and B, and
(2) Antibodies (Abs) for these alloAgs (called alloAbs) are regularly present in the serum of those individuals who lack the corresponding alloAg and are never in the serum of those who possess it.
The blood groups or types in human populations are named for the red cell Ags: group A has the A alloAg, group B has the B AlloAg, group O has neither, and in group AB each red cell has both A and B. The corresponding serum alloAbs are anti-A in group B persons and anti-B in group A; group O has both anti-A and anti-B; group AB has neither.
The ABO gene has three alleles – A, B, and O – with A and B dominant o 0. Since man is diploid, the two alleles per individual provide the six genotype and four phenotypes as follows:
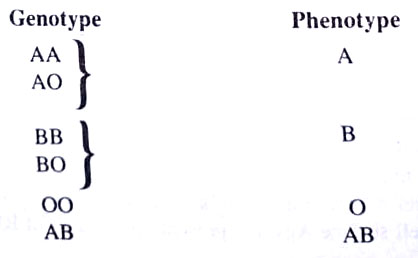
Four major blood types are thus known: A, B, AB, and O. Each type is distinguished by certain antigens on the surface of the erythrocytes and certain antibodies in the plasma.
Before transfusion, it is important that the blood types of participants are determined so that incompatible types are not mixed. For instance, if Type A blood donates to a recipient whose Type is O the A antigens on the donor’s erythrocytes will react with antibodies in the recipient plasma and the cytotoxic effect will be expressed as agglutination of donor erythrocytes in the recipient’s circulatory system.
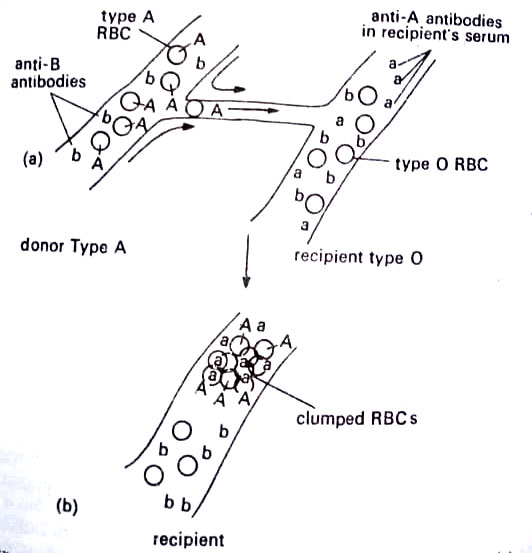
If the conditions are reversed, the donor’s A antibodies will react to a lesser degree with the recipient’s A antigens, because dilution in the recipient’s plasma takes place. The older notions that Type O blood is the universal donor and that type AB is the universal recipient are untenable. It is always desirable to cross-match the donor’s erythrocytes with the recipient’s plasma as well as the reverse, to ensure compatibility.
Another common expression of this type of hypersensitivity is hemolytic disease of the newborn or Rh disease. Erythrocytes of some persons contain an antigen first described in rhesus monkeys and therefore known as Rh antigen. These persons are said to be Rh-positive and those lacking it as Rh-negative. Infact the antigen is a group of antigens that vary among Rh-positive individuals.
However, here we will consider it as a single factor. Rh antigen-producing ability is an inherited trait. If an Rh-negative woman marries an Rh-positive man, there is a 3 to 1 chance or 75% probability that the trait will be passed to the child and that the child will be Rh-positive. Normally, a woman’s circulatory system is exposed to her child’s blood during the birth process, and if the child is Rh-positive, its antigens enter her blood and stimulate her immune system to produce antibodies.
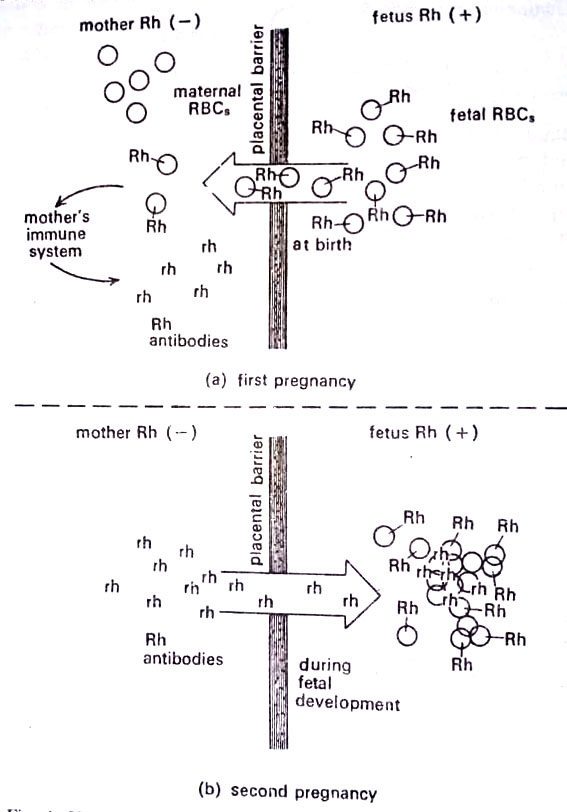
If succeeding pregnancy results in another Rh-positive child, these antibodies will cross the placentas (along with other antibodies) and enter fetal circulation. There they will react with the Rh antigens on the fetal erythrocytes and cause complement-mediated lysis of the cells. The fetal circulatory system rapidly releases immature erythroblasts to replace the Iysed blood cells, but these are also destroyed. From this observation, the old name of the disease is erythroblastosis fetalis. The result may be a stillbirth.
Modern into the women within 72 hours of delivery of an Rh-positive child. Antibodies in the preparation injected interact with Rh antigens and remove them from the circulation thereby preventing stimulation of the woman’s immune system. It should, however, be noted that an Rh-negative woman may produce rh antibodies as a result of the abortion of an Rh-positive fetus or after a transfusion with Rh-positive blood. (BSc Microbiology Immune Disorders Notes Study Material)
Other examples of cytotoxic hypersensitivity are some autoimmune diseases, that are not much familiar. These diseases are called autoimmune diseases since in them antibodies are directed toward the individual’s own cells. Some such diseases include (a) thrombocytopenia, resulting from antibodies produced against certain drugs such as aspirin, antibiotics, antihistamines, etc., (b) agranulocytosis, i.e. destruction of neutrophils by antibodies, also stimulated by drugs, and (c) Goodpasture’s syndrome, where antibodies combine with antigens on the membranes of glomeruli in kidneys.
Type III–Immune Complex Hypersensitivity
This type develops when antibodies combine with antigens and form aggregates that accumulate in blood vessels or on tissue surfaces. As complement is activated, the C3a and C5a components increase vascular permeability and exert a chemotactic effect on phagocytic neutrophils, drawing them to the target site. Here the neutrophils, release lysosomal enzymes that cause tissue damage. The antibodies are predominantly IgG, with IgM also found in some cases. (Microbiology Immune Disorders Notes Study Material)
Serum sickness is a common manifestation of this type of hypersensitivity. It develops when the immune system produces IgG against residual proteins in serum preparations. Other examples of this type, are the Arthus phenomenon, caused by antigens in the dust from moldy hay and in dried pigeon feces, and Systemic lupus erythematosus (SLE), an autoimmune disease in which B-lymphocytes produce IgG upon stimulation by nuclear components of disintegrating white blood cells.
Type IV-Cellular Hypersensitivity
This type is an exaggeration of the process of cellular immunity discussed earlier. There is contact between T-lymphocytes and antigens where lymphokines act as mediators. Lymphokines are a series of lymphoblast-derived proteins that increase the efficiency of phagocytosis of antigens. This type of hypersensitivity is a delayed reaction whose maximal effect is not seen until 24 to 72 hours have elapsed. It is characterized by a thickening and drying of skin tissue, a process called induration, and a surrounding zone of erythema (redness).
There are two major forms of this hypersensitivity: infection allergy and contact dermatitis.
[I] Infection allergy
It develops when the immune system responds to certain microbial antigens, such as bacterial agents of tuberculosis, leprosy, and brucellosis, the fungi involved in blastomycosis, histoplasmosis, and candidiasis, the viruses of smallpox and mumps, and the chlamydiae of lymphogranuloma. T-lymphocytes migrate to the antigen site and release lymphokines which attract phagocytes and encourage phagocytosis. An important application of infection allergy is the tuberculin skin test for tuberculosis.
[II] Contact dermatitis
It develops after exposure to a broad variety of antigens such as the allergens in clothing, jewelry, insecticides, coins, cosmetics, furs, etc. Other substances may include copper, formaldehyde, dyes, bacterial enzymes, protein fibers, etc.
The course of contact dermatitis is typical of a type IV reaction. Repeated exposures cause drying of the skin, with erythema and scaling. There are unlimited possibilities that cause this type.
Immune Deficiency Diseases
Such diseases may be life-threatening in some cases. Those, that are relatively minor deficiency states, may develop when there is malnutrition. Following are some examples of immune deficiency diseases. (BSc Microbiology Immune Disorders Notes Study Material)
- Bruton’s agammaglobulinemia. This was first described by O.C Bruton in 1952. Here B-lymphocytes fail to develop from pre-B-lymphocyte cells in the bone marrow. The lymphoid tissues lack plasma cells; levels of all five types of antibodies are low or absent in the patient, and there are no antibody responses. This is a sex-linked inherited trait more frequent in males than in females. Artificially acquired passive immunity is the treatment. (Microbiology Immune Disorders Notes Study Material)
- DiGeorge’s syndrome. Here T-lymphocytes fail to develop. The deficiency is linked to the failure of the thymus gland to mature in the embryo. Cellular immunity is defective in such individuals.
- Severe combined immunodeficiency (SCID). This is the most dangerous one, as it is accompanied by defects in both humoral and cellular immunity. The B-lymphocyte and T-lymphocyte areas of the lymph nodes are depleted of lymphocytes, and all immune function is suppressed. The disease develops due to failure in the normal development of both the thymus and bursa equivalent.
The longest surviving victim of SCID was a boy, David, who lived for 12 years inside a sterile, plastic bubble at the Baylor College of Medicine in Houston. In 1983, he received a bone marrow transplant from his sister for establishing his immune system. But later he died, when was removed from the bubble, as a victim of blood cancer, the virus brought through a transplant. - Chediak-Higashi syndrome. There is delayed killing of phagocytized microbes traced to the inability of lysosomes to release their contents.
- Job’s syndrome. This is accompanied by defective chemotaxis. The diseases (4) and (5) are linked to polymorphonuclear (PMN) cells. In this disease, phagocytes fail to engulf and kill microbes due to defects in chemotaxis, ingestion, and intracellular digestion.
- Acquired immune deficiency syndrome (AIDS). This is considered a viral disease. Here humoral immunity may also be altered. The number of T-lymphocytes is sparse. There is an imbalance between the helper T-lymphocytes and suppressor T-lymphocytes. Normally, the former type is twice in number that of the latter type. But in AIDS, the number of helper cells dramatically decreases.
Transplantation Immunology
Transplantation technology dates back to 1870 when J. Reverdin grafted bits of skin to wounded tissues. A kidney transplant was done in 1954 between identical twins. However, later attempts at kidney transplants were less successful.
During the next few decades, transplantation technology improved considerably. At present there exist four types of transplantations or grafts, depending upon the genetic relationship between donor and recipient. (BSc Microbiology Immune Disorders Notes Study Material)
- Autograft. A graft taken from one part of the body and transplanted to another part of the same body is called an autograft. This graft is never rejected since it is the person’s own tissue.
- Isograft. This is a tissue taken from an identical twin and grafted to another twin. This too is not rejected.
Rejection mechanisms become more vigorous as the genetic constitutions of donor and recipient cells become more varied. For example, grafts between brother and sister may lead only to mild rejection. However, grafts between cousins may be rejected more rapidly.
- Allografts. These are the grafts between random members of the same species such as two humans. These have variable degrees of success.
- Xenografts. These are grafts between members of different species such as monkeys and a human. These are rarely successful.
The transplanted tissue is rejected by the body the immune system interprets the tissue to be non-self. The rejection mechanisms may take two forms.
(1) Skin and tumor grafts are rejected mainly by the T-lymphocyte that enter the tissue and release lymphokines. Phagocytes are attracted to the site and the number of lysosomes in each phagocyte increases. Digestion of the tissue by phagocytes leads to drying and thickening as in Type IV hypersensitivity, and cell death or necrosis soon follows. (Microbiology Immune Disorders Notes Study Material)
(2) In kidney, heart, or other organ transplants, there occurs a different process. The blood vessels of the organ remain functional in the recipient’s body, and antigens stimulate host B-lymphocytes to produce antibodies. The antibodies enter the organ, where they combine with the antigens on the surface of the cells lining the vessel walls. This interaction activates complement, and the cells undergo lysis in a cytotoxic hypersensitivity. (Microbiology Immune Disorders Notes Study Material)
A rejection mechanism of a completely different type is sometimes observed in bone marrow transplants. In this case, the transplanted marrow may contain immune system cells that form immune products against the host after the host immune system has been suppressed during transplant therapy. Essentially the graft is rejecting the host. This phenomenon is called Graft Versus Host Reaction (GVHR).
Major histocompatibility complex (MHC). During the 1970s, immunologists could know that acceptance or rejection of graft depends largely on a relatively small number of genes called the major histocompatibility genes. Collectively, the genes are known as the major histocompatibility complex. This complex provides the code for a series of cell surface antigens called the histocompatibility antigens, also known as human leukocyte antigens (HLA) because they were originally studied in leukocytes. MHC in humans exists on chromosome number 6, close to immune response (Ir) genes. The complex consists of four clusters of genes, each gene having a number of versions.
MHC of genes and antigens is the key feature in the acceptance or rejection of a transplant, the closer the match between donor and recipient antigens, the more chance of a successful transplant. This is why tissue typing (a process used to determine how close two tissues match genetically) is used as a prerequisite to transplant surgery. (Microbiology Immune Disorders Notes Study Material)
Antirejection mechanisms. To arrest rejection it is often necessary to suppress the immune system. There are certain methods to do that. One method is the use of drugs. There are antimitotic drugs, which prevent the multiplication of lymphocytes in the lymph nodes. Such drugs are azathioprine, Cyclosporin A, etc. Another method includes the introduction of antibodies against the lymphocytes. There are also used some steroid hormones like prednisone. (Microbiology Immune Disorders Notes Study Material)
Tumor Immunology
The change from a normal cell to a cell with tumor potential is accompanied by several physiological changes. Tumor cells contain antigens not found in surrounding cells. These antigens may induce an immune response and make the cancer cells vulnerable to destruction. However, the cells escape the resistance mechanisms of the body and proliferate to form tumors. As the tumor breaks apart and spreads, metastasizing cancer develops. Four types of antigens have been identified in tumor cells.
One type, called oncofetal antigen occurs in certain tumors as well as in fetal tissues. An example is alpha-fetoprotein (AFP), an antigen found in embryonic development and several cancers, especially of the liver. An example is a carcinoembryonic antigen (CEA) also found in fetal tissues as well as cancer of the colon. The second type of antigen is found in tissues transformed by chemical carcinogens. The third and fourth types of antigens are those associated with DNA viruses and RNA viruses respectively.
Host resistance to tumors depends on immune responses directed against the tumor antigens. This process is termed immune surveillance. It is considered an ongoing function of killer T-lymphocytes which recognize the antigens as foreign and destroy the cells containing them in a way similar to graft rejection. Another cell thought to function in immune surveillance is the natural killer (NK) cell, which exerts a nonspecific, cytotoxic effect against other cells, including tumor cells. (Microbiology Immune Disorders Notes Study Material)
Immunologic escape and immunotherapy
In spite of immune surveillance, tumors occur in normal individuals because tumor cells escape destruction. Several theories are put to explain this immunologic escape.
For instance, (i) individuals learn to tolerate tumor cells before immune competence has developed and then express the tumor later in life, (ii) the majority of cancer cells are destroyed by killer cells, but that rapidly growing variant emerges and outpaces the immune system, (iii) that tumor antigens elicit antibodies that protect against the killer cells, (iv) that tumor cells release substances that suppress the immune system. None of these theories is universally accepted. (Microbiology Immune Disorders Notes Study Material)
Immunotherapy for the management of cancer is developing along several lines.
It includes (i) injection of the killer lymphocytes specific for the tumor, or monoclonal antibodies for tumor antigens injected. (ii) use of interferon to finance the activity of natural killer cells. In 1986, interferon was used against hairy cell leukaemia, cancer of white blood cells named for the hairlike appendages on malignant cells, (iii) use of interleukin 2, a T-lymphocyte protein, began in 1985.
This protein stimulates the rapid multiplication of the cell that produced it as well as other helper T-lymphocytes. (iv) the use of another anti-cancer agent, the tumor necrosis factor (TNF), a protein product of macrophages discovered in 1975. TNF is currently produced by genetic engineering and is shown effective against 20 kinds of cancer cells grown in culture.
BSc Microbiology Immune Disorders Notes Study Material
BSc 2nd Year Sample Model Practice Mock Test Question Answer Papers